Drug Resistance
Research shows that a growing number of yeast species are developing resistance to fluconazole 3
Yeast species are less resistant to miconazole, the
active ingredient in MONISTAT®
4 facts about drug resistance in VVC
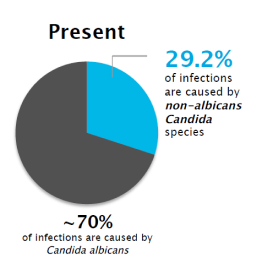
1The prevalence off VVC caused by non-albicans Candida 3
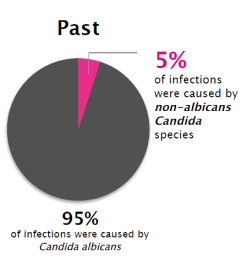
The prevalence of VVC caused by non-albicans Candida has increased greatly-and these species are less susceptible to fluconazole18
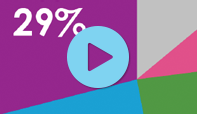
Although Candida albicans is still the most common cause, non-albicans species now account for 30% of vaginal yeast infections. Many are resistant to commonly prescribed antifungal agents, making treatment more challenging.
2Fluconazole resistance often begins in the gut
Oral fluconazole is systemic, and a significant amount of the drug remains in the GI tract. Repeated use reduces the C. albicans population in the gut, allowing resistant non-albicans species to thrive and reach the vagina.4
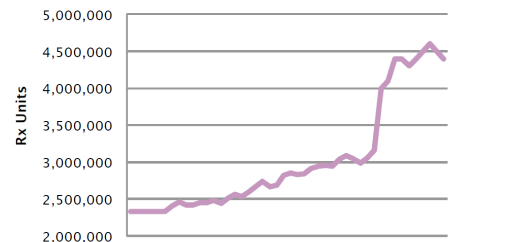
Source: IMS 2Q2016, IPSOS Survey August, 2016
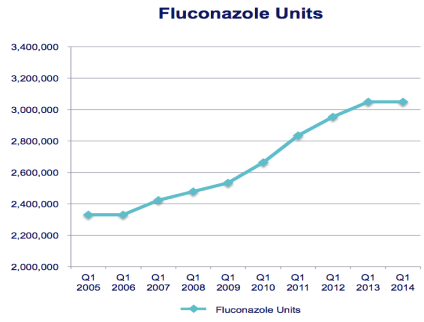
3Fluconazole doses have increased dramatically
The number of fluconazole pills prescribed has more than doubled since 2005, and nearly 60% of patients get more than 1 fluconazole tablet initially.5 These dosing trends may be another indicator of growing resistance.
4Optimal treatment for non-albicans VVC is not yet established
CDC Guidelines suggest a non-fluconazole azole for 7-14 days first-line2
The Guidelines also include recommendations for treating VVC in certain populations, including women who are pregnant, diabetic, or immunocompromised.
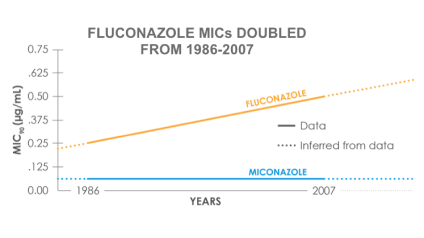
In a study looking at MIC90 trends from 1986 – 2007 for 250 C. albicans vaginal isolates3:
- Miconazole: MIC90 unchanged (0.06 μg/mL)
- Percent isolates with MIC90 ≥ 0.5 μg/mL increased from 2% to 9% during this period but since this is a topical agent and vaginal fluid levels exceed 10 μg/mL, resistance is unlikely to be an issue

Evolution and selection of vaginal-colonizing Candida 3
Evolution and selection of vaginal-colonizing Candida Candida species with reduced susceptibility could play a critical early role in the development of antifungal resistance among C. albicans isolates responsible for refractory candidiasis.3
- Fluconazole MIC90: 0.25 μg/mL → 0.5 μg/mL
- Percent isolates with MIC90 ≥ 1 μg/mL and ≥ 2 μg/mL both increased from 3% to 9% over this period
- While not a clinically significant MIC90 increase, the increase in isolates with elevated MIC90 may have clinical relevance given the achievable concentrations of fluconazole in vaginal fluid (maximum 2 μg/mL)
Fluconazole use has increased dramatically over time3
Routine use of prescription oral fluconazole may create an environment where non-albicans Candida, which are less susceptible to azoles, thrive and spread in the vagina.
- Oral fluconazole achieves low tissue concentration at the site of infection while the rest remains in systemic circulation.
- A significant amount of the drug remains in the GI tract, reducing C. albicans but allowing less-susceptible non-albicans species to grow.
- These non-albicans species reach the vagina and are more likely to be resistant to fluconazole treatment.
Repeat systemic antifungal therapy perpetuates drug-resistant species.